Heidelberg, 11 March 2024. It’s not every day that we get an intraocular lens (IOL) made of a new material!
Today, we are very pleased to see a preview of our paper to be published in the April edition of the Journal of the Mechanical Behavior of Biomedical Materials; (and online since 2nd January.)
In-vitro assessment of a novel intraocular lens made of crosslinked polyisobutylene Sonja K Schickhardt, Grzegorz Łabuz, Donald J Munro, Ingo Lieberwirth, Lu Zhang, Hui Fang, Gerd U Auffarth.
2024 marks 75 years since Sir Harold Ridley’s IOL operation in 1949.
Ridley’s choice of IOL material was PMMA (polymethylmethacrylate). Later, other materials were used in IOL manufacture, chosen for their foldability - silicone elsatomers, hydrogels and acrylates. Today, we have the Eyedeal® lens, made of a new material - cross-linked polyisobutylene (xPIB).
What we did
We assessed the material quality using an accelerated ageing process (to provoke glistenings) and compared values with a control, the widely implanted and successful AcrySof® lens.
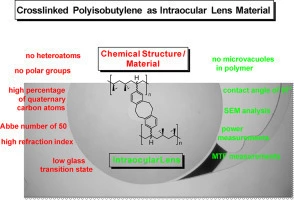
Using the sessile drop method, we measured the contact angle of the new IOL. We recorded images of the lens surface by scanning electron microscopy (SEM).
Using our Lab’s standard metrology device (OptiSpheric IOL PRO2) we assessed Optical quality by measuring the labeled power and modulation transfer function (MTF)).
What we found
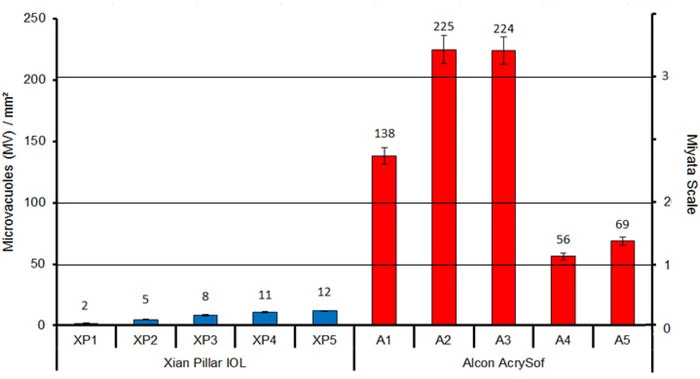
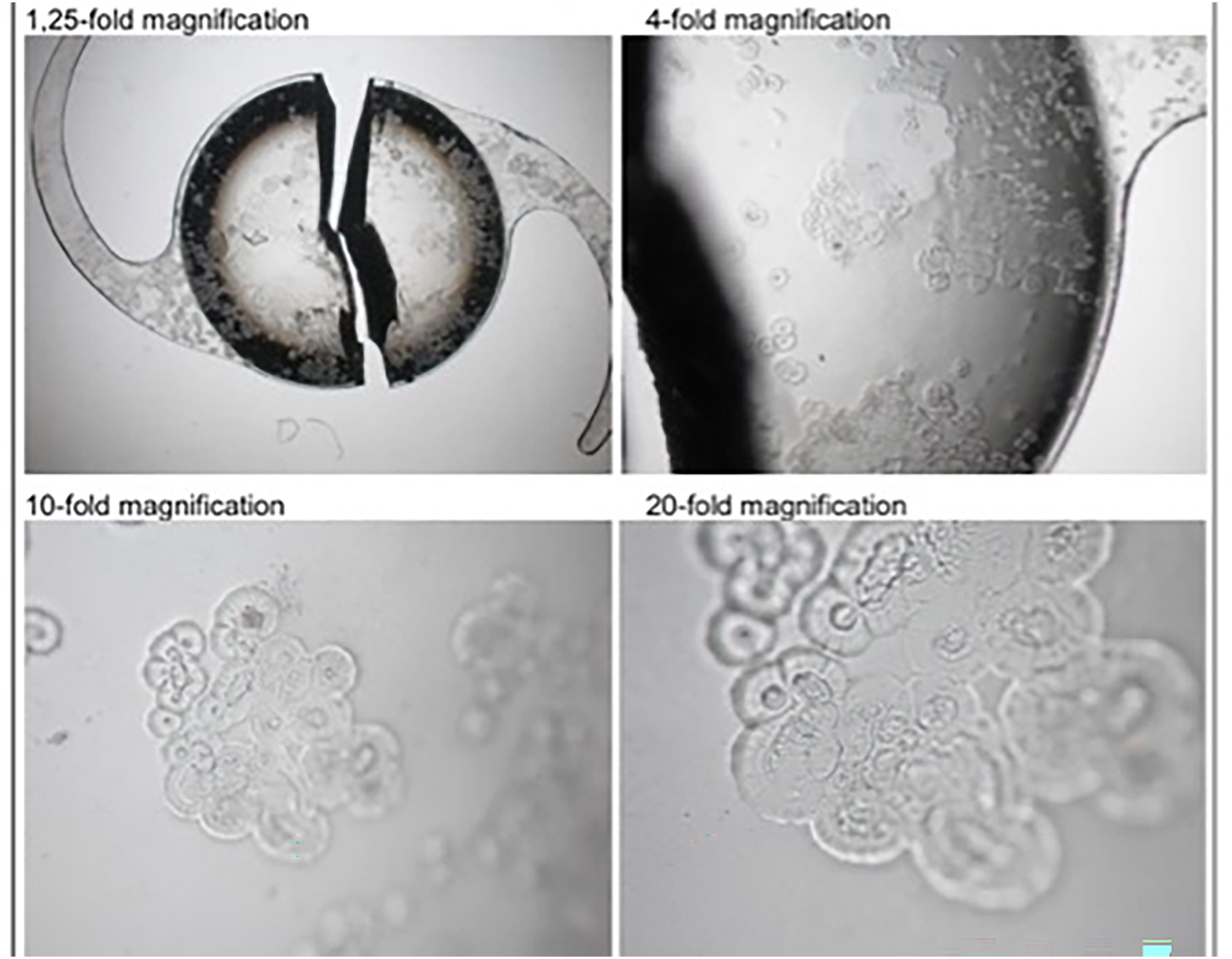
- The Eyedeal® lens had an average glistening density result of 7.46 ±3.78 MV/mm2 compared to the control AcrySof® whose glistenings number was 142.42 ±72.47 MV/mm2.
- The contact angle was 97.2◦whereas the angle of AcrySof material is between 73.3 ±2.4◦ and 84.4 ±0.1◦.
- Using SEM, all Eyedeal® lenses and appeared to be comparable to modern IOLs made of acrylic materials.
- The power and MTF values were normal and conformed to ISO standards.
In a nutshell
Although its use in IOL manufacture is new, cross-linked polyisobutylene already has a good track record in the manufacture of other medical devices including a glaucoma device. Clinical studies on the xPIB IOL have already commenced in China and will come to Europe this year. In our laboratory, the new Eyedeal® lens showed equivalence to current hydrophobic- or hydrophilic-acrylic lens models. The Eydeal lens showed superiority in its glistening density result compared to the Acrysof control lens.
Enjoy our paper, it is an open access article, available online:
In-vitro assessment of a novel intraocular lens made of crosslinked polyisobutylene - ScienceDirect



 Team VitreoSub is a winner!
Team VitreoSub is a winner!