Heidelberg, 5 February, 2021. We are pleased to report that our latest research paper is published today in the Ophthalmology and Therapy journal: Quantification of the In Vitro Predisposition to Glistening Formation in One Manufacturer’s Acrylic Intraocular Lenses Made in Different Decades.
Why study this?
We know that foldable hydrophobic acrylic intraocular lenses (IOLs) are sometimes prone to develop a long-term postoperative material change called Glistenings. In the laboratory, we can induce glistenings. We aimed to investigate the hydrophobic acrylic IOL material from one manufacturer - Alcon, Fort Worth, TX, USA - and from different years of manufacture, from the introduction of the AcrySof lens in 1990 to the present day Clareon, in each case looking for changes in the lens material’s predisposition for glistening formation. We hypothesized before starting the study, that the tendency toward glistening formation has decreased over the last thirty years, from the 1990s until today.
What did we do?
In our laboratory in Heidelberg, we have access to the late David J Apple’s archives, all the paper records of the explant and autopsy IOLs that he received since he set up his Lab in 1980. But the record is not just documents - often it includes explanted lens specimens. For our study we were interested in the explanted AcrySof lenses (Alcon, Fort Worth, TX, USA). We chose lenses sent to the Lab in 1996 and 1997. These were sent by surgeons in Italy, Canada and the majority from USA to the Lab - then in Charleston, South Carolina. We examined twenty-three explant specimens from this historical resource - AcrySof three-piece IOLs (MA30BA/MA60AC).
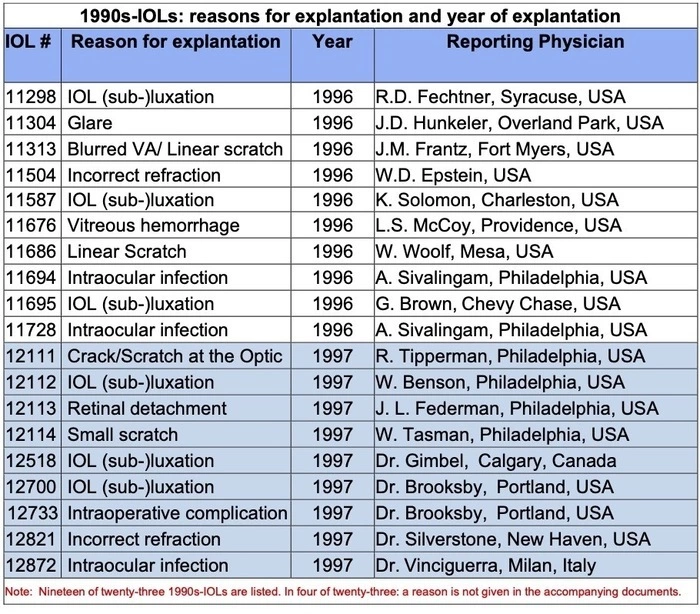
More than twenty years later, several of the surgeons in this list are still active in ophthalmology or have only recently retired. Pictured while visiting Charleston, the young Canadian surgeon Howard Gimbel sits at the microscope of Prof. Apple’s Miyake-Apple table. (Dr Gimbel is known for his co-invention, with Thomas Neuhann from Munich, of the continuous curvilinear capsulorhexis (CCC) technique.) We thank Dr Gimbel and other surgeons for 23 or 24 years ago sending these specimens. (In the 1990s surgeons were aware of Glistenings. But we may remark that in none of our series of explanted 1990s-IOLs is glistening expressly given as the reason for explanting the lenses. In all our cases, the lens implant was only a few months in the eye or removed a few days after surgery - too soon perhaps for Glistenings to develop.)
Pictured while visiting Charleston, the young Canadian surgeon Howard Gimbel sits at the microscope of Prof. Apple’s Miyake-Apple table. (Dr Gimbel is known for his co-invention, with Thomas Neuhann from Munich, of the continuous curvilinear capsulorhexis (CCC) technique.) We thank Dr Gimbel and other surgeons for 23 or 24 years ago sending these specimens. (In the 1990s surgeons were aware of Glistenings. But we may remark that in none of our series of explanted 1990s-IOLs is glistening expressly given as the reason for explanting the lenses. In all our cases, the lens implant was only a few months in the eye or removed a few days after surgery - too soon perhaps for Glistenings to develop.)
We also examined five of the newer AcrySof IOL models (MA60AC, SA60AT, TFNT00 and SN60WF) that were manufactured between 2014 and 2017.
Finally, we looked at the company’s newest model, and we put five Clareon (SY60WF) IOLs through the same accelerated ageing procedure. We counted the number of microvacuoles per square millimetre (MV/mm2) in the central part of each IOL optic, and we compared this number between the groups.
What we found
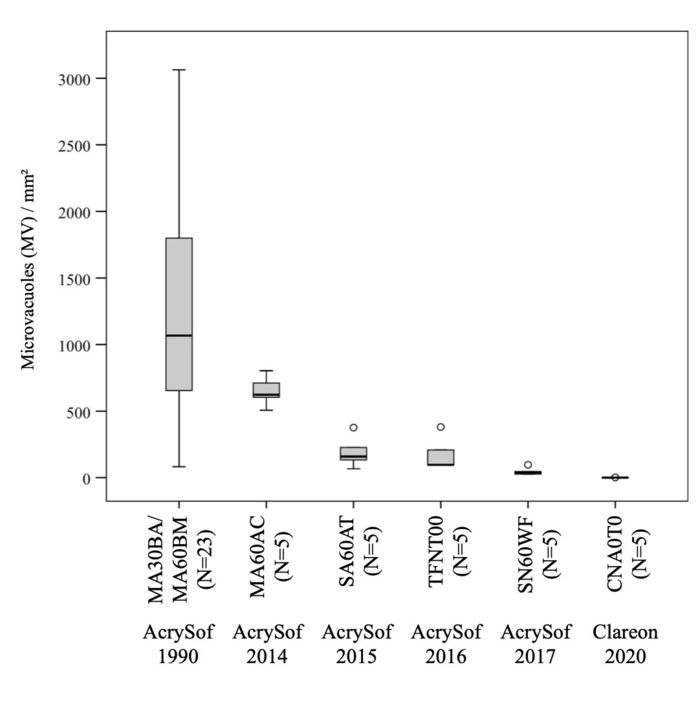 The mean number of microvacuoles was highest in the 1990s-manufactured Alcon AcrySof IOLs, with 1289 (± 738) MV/mm2. This number decreased to 650 (± 101), 192 (± 105), 175 (± 112) and 47 (± 26) for MA60AC, SA60AT, TFNT00 and SN60WF, respectively made in the second decade of this century.
The mean number of microvacuoles was highest in the 1990s-manufactured Alcon AcrySof IOLs, with 1289 (± 738) MV/mm2. This number decreased to 650 (± 101), 192 (± 105), 175 (± 112) and 47 (± 26) for MA60AC, SA60AT, TFNT00 and SN60WF, respectively made in the second decade of this century.
The lowest count was in the new Clareon IOL, with 1 (± 1) MV/mm2.
Continual improvement
We could induce a high number of glistenings in the specimens of explanted IOLs from the 1990s. The propensity for glistening formation decreased considerably after that decade and even though in vitro glistening formation in today’s AcrySof lens material was low, the new Clareon material was found to be the best: essentially glistenings-free.
Our study confirms the manufacturer’s own accounts (given at different times) that changes in lens packaging and (however, not defined) improvements in the production process have decreased the material’s propensity to develop glistenings.
Read and download the peer-reviewed publication
Ophthalmology and Therapy Journal.
This journal is part of the Springer Group of publications, and it has a growing reputation not least for its Rapid Publication procedures, conducting a rapid peer review of 2 weeks and typically once it is accepted, it will publish within a few weeks.From the Springer website:
The rapid timelines are achieved through the combination of a dedicated in-house editorial team, who manage article workflow, and an extensive Editorial and Advisory Board who assist with peer review. This allows the journal to support the rapid dissemination of research, whilst still providing robust peer review. Combined with the journal’s open access model this allows for the rapid, efficient communication of the latest research and reviews, fostering the advancement of ophthalmic therapies.
Cite this article
Wang, Q., Yildirim, T.M., Schickhardt, S.K. et al. Quantification of the In Vitro Predisposition to Glistening Formation in One Manufacturer’s Acrylic Intraocular Lenses Made in Different Decades. Ophthalmol Ther (2021). https://doi.org/10.1007/s40123-020-00329-8



 Reversible Trifocality
Reversible Trifocality