 Contributed by Dr. Hyeck-Soo Son. Heidelberg 01 August 2020. We are very pleased to see our research paper published in the Journal of Cataract and Refractive Surgery, indeed the journal shows images from our report on the front cover - in the top row one can see the patient’s eye before Artificial Iris surgery and after surgery. The lower photographs on the cover, show the high level of aesthetic matching of the operated (left) eye with the unoperated right eye.
Contributed by Dr. Hyeck-Soo Son. Heidelberg 01 August 2020. We are very pleased to see our research paper published in the Journal of Cataract and Refractive Surgery, indeed the journal shows images from our report on the front cover - in the top row one can see the patient’s eye before Artificial Iris surgery and after surgery. The lower photographs on the cover, show the high level of aesthetic matching of the operated (left) eye with the unoperated right eye.
In vitro optical quality assessment of a monofocal IOL sutured to an artificial iris. Christian Mayer, MD, Hyeck-Soo Son, MD, Grzegorz Łabuz, Ph.D., Timur M. Yildirim, MD, Gerd U. Auffarth, MD, Ph.D., Ramin Khoramnia, MD J Cataract Refract Surg 2020; 46:1184–1188
What was the research?
We already know that we can safely combine implantation of a monofocal IOL and an artificial iris to treat patients with aphakia and aniridia. But we wanted to assess whether the combined implantation affects the IOL’s optical performance.
What did we do?
We made an in vitro laboratory study at the David J. Apple International Laboratory for Ocular Pathology, using the OptiSpheric IOL Pro II device to measure the IOL’s modulation transfer function (MTF) at 3.0 mm pupil size and spatial frequency of 100 lp/mm. We used three ASPIRA®-aAY IOLs with different base powers, 10 diopters (D) (IOL A), 20D (IOL B), and 30D (IOL C) - measured before and after suturing the IOL to an ArtificialIris (AI). We also looked at the degree of IOL decentration about the center of the AI.
The Artificial Iris
The Artificial Iris (AI) we used was the ArtificialIris®, made by HumanOptics, Erlangen, Germany. The CE mark, Conformité Européenne certificate, was granted to the ArtificialIris in 2011, and it was approved by the US Food and Drug Administration in the United States in 2018.
It is designed for posterior segment implantation in aphakic and pseudophakic eyes. The AI can be implanted alone or combined with an IOL. It has a 12.8 mm overall diameter with a fixed pupil size of 3.35 mm. Its thickness decreases from the pupillary margin (0.4 mm) to the periphery (0.25 mm). The AI is made from a biocompatible silicone material that has a black posterior surface, which is optically-opaque, and a colored anterior surface. This anterior surface is a hand-painted reproduction of the patient’s iris, taken from a photograph of the patient’s residual iris tissue (RI) and the fellow eye’s iris.
A high-resolution photograph needs to be obtained from both eyes, which is then sent to the manufacturer for the implant to be individually hand-painted. By incorporating colored silicone pigments in various layers, the manufacturer creates a three-dimensional plastic surface that resembles the natural iris surface with its crypts as well as giving a natural look that minimizes light reflections.
 The images A to F show the process of attaching with sutures the AI to the IOL. The IOL is sutured to the posterior side of the Artificial Iris.
The images A to F show the process of attaching with sutures the AI to the IOL. The IOL is sutured to the posterior side of the Artificial Iris.
What did we find?
Below, the three images of the decentring of each IOL. The decentration values in vertical direction were 0.20 mm, 0.00 mm, and 0.02 mm for IOLs A, B, and C, respectively.
 In the horizontal direction, the decentration values were 0.42 mm, 0.10 mm, and 0.03 mm for IOLs A, B, and C, respectively.
In the horizontal direction, the decentration values were 0.42 mm, 0.10 mm, and 0.03 mm for IOLs A, B, and C, respectively.
The mean MTF values before suturing were 0.57, 0.65, and 0.63 for IOLs A, B, and C, respectively. After suturing to the AI, the mean MTF values were 0.52, 0.54, and 0.55 for IOLs A, B, and C, respectively. 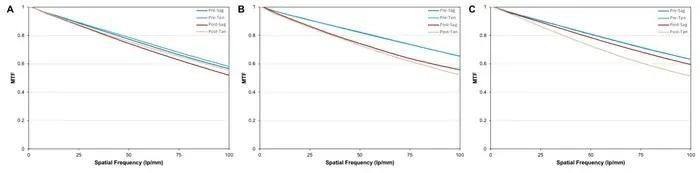
The MTF decreased slightly in all 3 IOLs after they were sutured to the AI. The small differences, however, should be clinically irrelevant. This laboratory assessment showed that the suturing of the IOL to the AI can be performed in a reliable and reproducible manner without deteriorating optical quality.
What was known
Combined implantation of a monofocal IOL and an artificial iris has become a safe and routine treatment for patients with aphakia and aniridia. However, we didn’t know whether the IOL’s optical quality deteriorates when it is sutured to the artificial iris.
What our paper adds
The optical performance of the monofocal IOLs deteriorated slightly after they were sutured to the ArtificialIris. However, given the generally excellent optical quality of monofocal IOLs, these changes are considered to be of no clinical relevance.



 Anneke Worst (1928-2020)
Anneke Worst (1928-2020)